
What we stand for
Quantoom Biosciences is a full-stack RNA partner for mRNA- and saRNA-based vaccines and therapeutics. Its N-Force toolbox relies on 3 core elements to turn any antigen into a (sa)mRNA-LNP drug product: Ncode for sequence design and optimization, Ntensify® for RNA production and Ncapsulate® for RNA-LNP formulation. Beyond technology, Quantoom Biosciences assists its partners by providing extensive enabling solutions, ranging from strategic R&D partnerships to sequence design & optimization.
Our expertise & offering
Quantoom Biosciences accelerates the transition towards RNA-based therapeutics. Through its end-to-end N-Force toolbox, it enables its clients to turn any antigen into a high-performing sa/mRNA-LNP drug candidate. It relies on three complementary pillars — Ncode, Ntensify®, and Ncapsulate® QCX — to design, produce, and formulate RNA ready for preclinical or clinical studies. Our Ncode platform maximizes the potential of any antigen by optimizing the nucleic acid sequence, the Ntensify product line provides a scalable and construct-agnostic RNA production and purification platform, and the Ncapsulate lipid library enables RNA formulation for de-risked therapeutics. Taken together, our N-Force toolbox offers the best end-to-end solution for any organization looking to transition to RNA-based therapeutics.


Expertise in Sequence design & optimization for sa/mRNA
We transform any antigen of interest into a fully optimized sa/mRNA construct, using a proven combination of design tools and scientific know-how. We guide our partners from the amino acid sequence to an optimized DNA template, ensuring that each construct is optimized for DNA manufacturing, RNA stability, and protein expression. Whether it is for infectious diseases through mosaic antigen designs or for oncology applications with mini-gene constructs, we have built extensive experience through our numerous partnerships with top-tier institutions such as the University of Arizona, Memorial Sloan Kettering Cancer Center, and Vaxthera. Our track record of successful designs has given us the capabilities to help accelerate and de-risk RNA development from the very first step.

Expertise in integrated and scalable RNA manufacturing
We have developed Ntensify as a fully integrated, scalable solution that redefines how RNA is produced. By combining process intensification, advanced design principles, and years of optimization, we enable high-yield RNA manufacturing with a drastically reduced footprint, fewer manual steps, and improved product consistency. Ntensify is designed to be construct-agnostic, easy to implement, and seamless to scale—from early R&D quantities to commercial-scale production. This approach reduces costs, increases reliability, and supports the broader availability of life-saving biologics.
The Ntensify process combines IVT and purification to streamline operations. The IVT step uses co-transcriptional capping with TriLink’s CleanCAP and has been optimized to produce minimal impurities, facilitating downstream purification. This optimization enables a transition to a single-step purification method based on magnetic silica beads, replacing the multiple conventional chromatographies and TFF steps typically required for bulk purification.
We provide a complete range of Ntensify platforms designed to support every stage of RNA development, from early construct assessment at R&D scale to clinical and commercial production. Our product line includes the Ntensify® mano, micro, mini, and midi systems, offering flexible capacities, scalable outputs, and increasing levels of automation—allowing partners to efficiently produce mRNA or saRNA from microgram to gram scale.



Expertise in high-performance RNA formulation
We developed delivery chemistries — Ncapsulate® QCX — designed to enhance mRNA and saRNA delivery across a wide range of in-vivo applications, drawing on deep formulation know-how and years of in-species validation to create lipid-like systems that consistently increase protein expression where it matters.
Keeping therapeutic applications in mind, we tailored our kits for in-vivo use with customized chemistries for intravenous and intramuscular administration, while also offering solutions for in-vitro R&D for both mRNA and saRNA.
We offer Ncapsulate QCX as a flexible delivery solution available in multiple formats to support a wide range of RNA programs.
- Ncapsulate® QCX core – our raw material enables clients accustomed to formulation to rapidly adopt our core lipid and start delivering RNA where it matters.
- Ncapsulate® QCX combo – our ready-to-formulate kits for rapid evaluation or fully customized LNP development for specific delivery needs.
- Ncapsulate® QCX carrier – our ready-to-use pre-encapsulated particles that can be formulated using simple mixers, without ethanol and need for downstream TFF.
- Ncapsulate® QCX custom – seeking a different solution compared to our off-the-shelf offering, we are geared to tailor our lipid, lipid mix and manufacturing process.
Our offering is designed to adapt to your preferred workflow—off-the-shelf, custom-configured, or raw material supply—ensuring reliable and scalable RNA formulation from early research through advanced development, while maintaining a solid IP position and a clear path to commercialization.
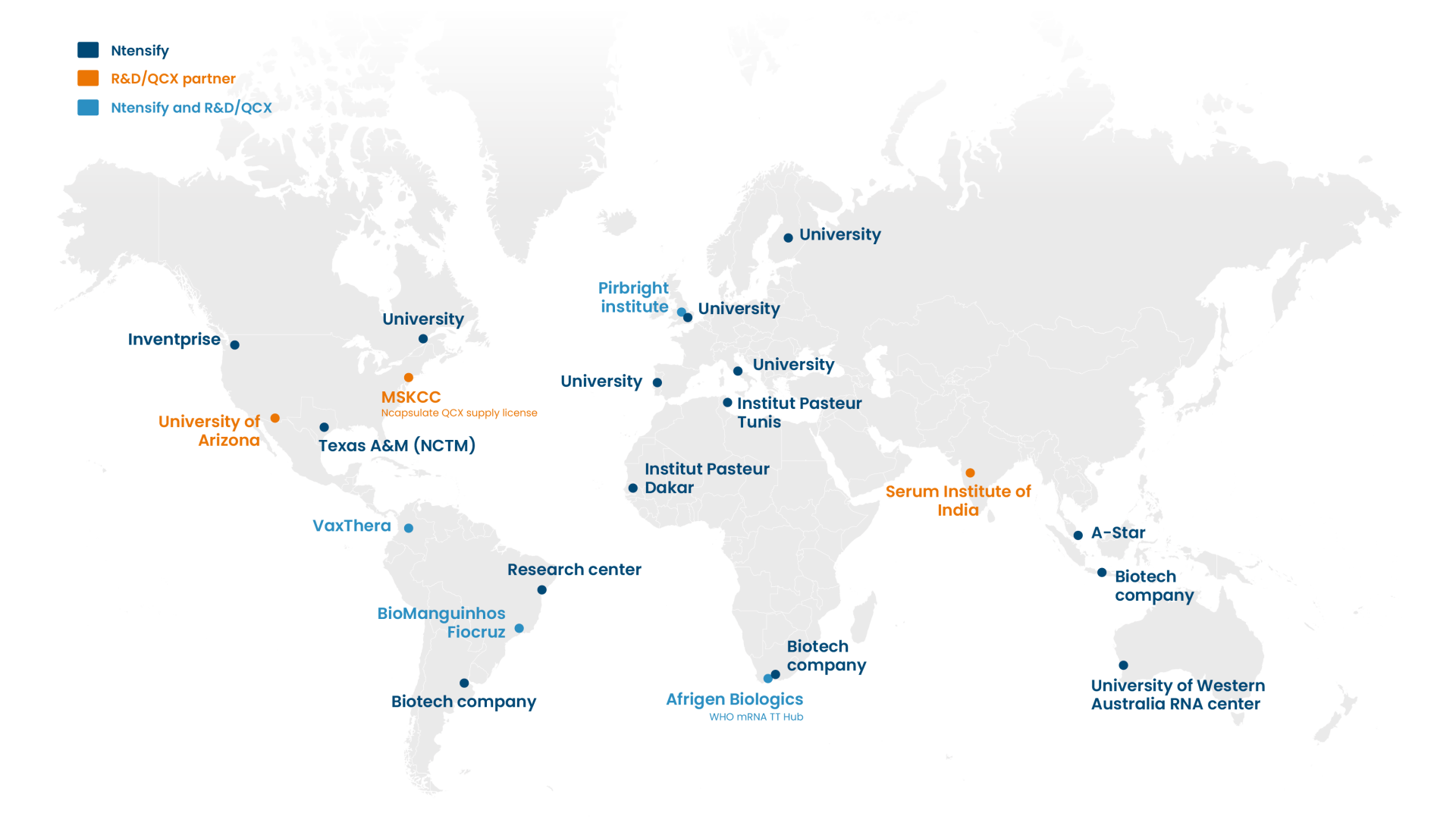
Our partners
Our global collaborations
Since its commercial launch in 2023, Quantoom Biosciences has installed 25 Ntensify® systems worldwide and has five ongoing R&D collaborations.
Diversity & values
At Quantoom Biosciences, we stand for inclusivity, diversity, and respect for individual differences. We strive to practice equality across the board, from our hiring methods to how we pay employees. Our team represents many diverse backgrounds and perspectives.
Accountability
We own and assume accountability for results, we establish high-performance standards
Continuous improvement
We challenge the status quo, embrace innovation, add value, and deliver operational excellence
Teach/lead
We value and solicit new ideas, develop and refine competencies by sharing knowledge and experience
Integrity
We maintain high ethical standards; respect, include and recognize differences
Team
Everyone acts as a team builder, we remove the barriers that block the team’s success
Our team
Management Team
Click on each picture to see the bio of each member

Jose Castillo, PhD.
Chief Executive Officer

Rachana Talwar
Chief of staff & Chief Operating Officer

Ashiqul Haque, PhD.
Chief Scientific Officer

Coralie Pirlot
Chief Financial Officer

Hala Audi
Global Director Alliance & Partnership

Noémie Jacquart
Head of Marketing & Sales Operations

Youlia Serikova, PhD.
Head of N-Force Process

Laurent Heusgens
Head of N-Force Product

Maria Davila-Saignol
Global Director Sales & Business Dev.
Key experts
Our team brings deep expertise across a broad range of capabilities
Our history
Quantoom Biosciences is a full-stack RNA partner for mRNA- and saRNA-based vaccines and therapeutics. Built around our N-Force toolbox — Ncode for sequence design, Ntensify® for scalable RNA production, and Ncapsulate® for RNA-LNP formulation — our platform turns any antigen into a high-quality (sa)mRNA-LNP drug product. Beyond technology, Quantoom Biosciences supports partners with extensive enabling services, from strategic R&D collaborations to sequence design and optimization.
University of Western Australia selects Ntensify® mini for advanced RNA research
The University of Western Australia’s RNA Innovation Foundry selects Quantoom Biosciences’ Ntensify® mini platform to support cutting-edge RNA research and technology development. This adoption highlights Quantoom’s growing footprint in academic research and innovation ecosystems, and its role in enabling the next generation of RNA-based therapies.
Restructuring and creation of Phoenix Biosciences as a standalone company
Quantoom Biosciences enters a new phase under the legal entity Phoenix Biosciences SA, through which all activities and technologies now continue. This restructuring provides a clear and solid foundation for the future, strengthening our position as a full-stack RNA solutions provider.
Research collaboration with Leading Global Cancer Center
Quantoom Biosciences signs a research license and supply agreement with Memorial Sloan Kettering Cancer Center to support the development of personalized mRNA-based cancer therapeutics. This collaboration confirms Quantoom’s expansion into high-value clinical research and oncology applications, reinforcing the versatility of its mRNA formulation and manufacturing technologies.
Launch of the Ntensify micro
Quantoom Biosciences launches the Ntensify® micro, expanding the R&D segment of its Ntensify® product line. The Ntensify® micro is the smallest and most compact system designed for the Ntensify® process. This benchtop unit enables the production and purification of 500 µg to 100 mg of high-quality mRNA in just 4 to 6 hours.
Belgium and Brazil unite to advance RNA-based therapies
Quantoom Biosciences signs a Memorandum of Understanding with Bio-Manguinhos/Fiocruz, strengthening international cooperation to accelerate the development and local production of RNA-based therapeutics. This milestone reflects Quantoom’s growing role as a global technology enabler, supporting sovereign manufacturing strategies beyond vaccines.
Strategic collaboration with Serum Institute of India
Quantoom Biosciences enters into a strategic collaboration with the world’s largest vaccine manufacturer to expand the accessibility of RNA-based personalized oncology and immunotherapies. This partnership marks a major step beyond vaccines, demonstrating Quantoom’s ability to support point-of-care, small-scale mRNA manufacturing for advanced therapeutic applications worldwide.
Launch of the Ntensify mano
Quantoom Biosciences announces the extension of the Ntensify product line with the launch of Ntensify mano, an mRNA kit containing mixes of reagents for mRNA production and purification for research use.
Installation of the 1st Ntensify midi
Quantoom Biosciences announces the installation of its first Ntensify midi system for mRNA vaccine production in the GMP facility of Afrigen Biologics in Cape Town, South Africa. (under 19 months of dev…)
Agreement for the first African-owned Covid-19 vaccine
The collaboration with Afrigen Biologics (hosting the World Health Organization’s Global mRNA Vaccine Technology Transfer Hub) will focus on the development of a novel mRNA vaccine that will drastically accelerate access to vaccination by using Quantoom’s mRNA production technology.
Collaboration with eTheRNA
Quantoom entered into a strategic collaboration to build an advanced, small footprint technology platform for the affordable production of RNA-based therapies. This platform was designed to be used either within existing facilities or rapidly deployed to areas of urgent need.
Official launch of Quantoom Biosciences
The COVID-19 pandemic underlined the desperate need for technological innovation in manufacturing to provide a simple, scalable way to make RNA. By lowering the barriers to RNA production, we allow our partners that have previously not considered exploring RNA-based vaccines and therapeutics, to gain access to RNA production capabilities and enter the market.







