RNA Production Solution
From R&D to large-scale manufacturing


Next generation
RNA production
The Ntensify RNA production solution is more than just equipment, it encompasses:
- A redesigned and optimized process
- Ready-to-use mixes of reagents
- Scale-less disposables
- Compact production equipment
This solution enables the synthesis and purification of messenger RNA with construct-agnostic performances. The technology is designed to maximize yield, quality, and cost-effectiveness, starting from a linear DNA template. The process produces purified naked RNA, ready for sterile filtration prior to formulation into a drug product.
Redesigned and
optimized process
The Ntensify solution is a novel approach to RNA production, achieved through advanced Design of Experiments (DoE) and years of optimization. It enables high-yield, low-contaminant RNA to be produced while simultaneously reducing the overall costs.
The process is construct-agnostic and includes an optimized one-pot IVT with co-transcriptional capping and single-step purification to minimize RNA losses and increase available drug-substance RNA. It can scale seamlessly from mg-scale (R&D level) to kg-scale (commercial production).
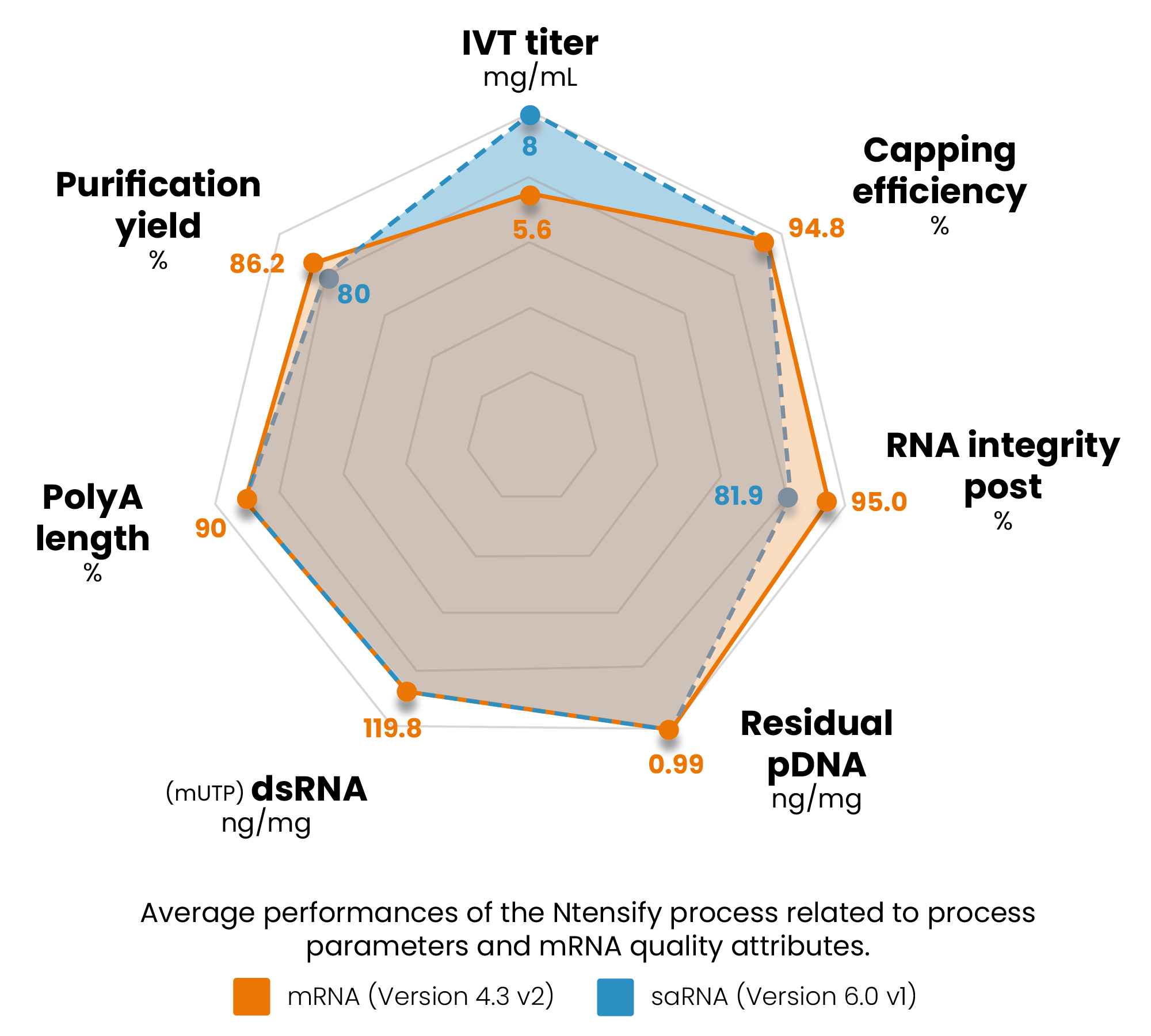
Cost-effective production of RNA drug substance
Quantoom Biosciences’ Ntensify process* has been developed through numerous experiments using various RNA constructs
- > 2000 individual mRNA reactions
- > 500 individual saRNA reactions
- 100+ different constructs tested
- Different mRNA sizes (1 to 4kb)
- Different saRNA sizes (8 to 12kb)
* Developed in collaboration with
Co-transcriptional capping
Helping you to shift from post- to co-transcriptional capping
To benefit from a simpler, more streamlined reaction process without additional purification steps.
Robust & performant process
- As shown by the 45 different constructs tested to date.
- High synthesis yield: 5,6 mg/mL.
High-quality mRNA
- 94,8% capping efficiency on average.
- 100% of 5’ cap 1 structure.
Simpler & faster process
Shorter timelines by incorporating a one pot IVT with co-transcriptional capping instead of a separate IVT and post-transcriptional capping
Cheaper
60% less capping reagent consumed compared to standard post-transcriptional capping with purification step.
Modified nucleotides
Offering maximum flexibility in the design of your drug candidate
The choice of uridines in mRNA sequences is crucial in drug research and development. Different uridine modifications affect the immune response, protein translation efficiency and vaccine stability. Optimizing uridine selection can enhance the effectiveness and safety of mRNA-based drugs.

Uridine
Endogenous variant with high immunogenicity

Pseudouridine
Improved variant with reduced immunogenicity

Methylpseudouridine
Stable mRNA with lower immunogenicity
One-step purification
Reducing the number of purification steps to increase recovery and drug substance availability
The purification process has been streamlined to a single step separation using silica-based magnetic beads, which is automated within the equipment following the IVT reaction. This reduces the number of purification steps, increasing recovery and drug substance availability. Not only does this minimize product losses, but it also decreases handling and the need for acquiring and validating various purification equipment.
Most importantly, this simplified method maintains stringent purification criteria, ensuring low residual contaminants in the final product.

Download the general plasmid design guidelines
For mRNA Ntensify process

For saRNA Ntensify process

Our product line
Our product line provides a complete solution for drug-substance RNA production, at every stage of the drug development process. Using the same design principles, our technology enables small scale experiments at milligram scale, all the way to commercial production of several hundreds of grams per batch.
Mixes of reagents
Our mixes of reagents are sourced from qualified vendors and guaranteed to be free of animal-derived components. These mixes are produced using a robust manufacturing process and undergo strict quality assurance testing before release. The quality of our raw materials remains the same at every scale, from Ntensify mano to Ntensify maxi. And all our mixes are supplied with CoA and SDS, TSE and BSE certificates. For GMP production, additional certificates for bioburden, endotoxins and RNAse are provided.
Our mixes of reagents mixes are supplied ready-to-use, eliminating the need for buffer preparation and ensuring process consistency. Through extensive development, our team has produced the best possible combination of enzymes, buffers, and other raw materials to maximize efficiency and reduce overall costs.
Ready-to-use mixes of reagents
The kit contains all necessary reagents for IVT and purification, pre-formulated at optimal concentrations in their final containers. This ready-to-use format ensures convenience and consistency in your workflow.
- Nucleotides
- Cap analog
- RNA-polymerase
- IVT Buffer
- DNAse
- Quenchers
- Purification beads & buffers
- Elution & Washing buffers
Our mixes of reagents are supplied ready-to-use with the following certificates:
- CoA
- TSE BSE
- SDS
- AOF
- RNAse
- Bioburden
- Endotoxins


67% CoGS Savings*
Spread across reagents and disposables
* Savings for an annual production of 100 million doses
The Ntensify process significantly reduces reagent consumption through enhanced IVT performance and simplified purification. By using a single equipment, it is possible to minimize the use of disposables. These improvements substantially reduce the cost of goods (CoGs) compared to conventional processes.
Small footprint facility
The Ntensify system’s compact footprint significantly reduces the required floorspace in both production suites and buffer preparation areas.
- No buffer proparation zone required
- IVT & DSP zones merged due to integrated equipment
- Global floorplan reduced by a factor 3
RNA production facility
with conventional equipment
RNA production facility
with Ntensify system
Proprietary
Disposables
The key to scalable RNA production lies in our proprietary disposables. Our IVT reactors address the challenges of conventional manufacturing scale-up via an automated scale-out approach, which significantly enhances efficiency, and reduces the complexities typically associated with scaling up RNA manufacturing.
Scalable IVT Reactors
Without the hassle of conventional scale-up.
Our disposables enable a remarkable leap in production capacity. These IVT reactors enable batch-continuous operation equivalent to multiple litres of IVT reaction, achieved through a scale-out approach starting from just 20mL reactions. This innovative design allows for the production of up to 150g of mRNA, equivalent to 3 million doses of a 50µg mRNA vaccine. Using the same process design principles, production can be scaled by a factor of 150 without the need to invest time and resources in scalability studies.


Pooled purification
To increase drug substance availability
To purify RNA while maintaining high recovery, the contents from various reaction cavities are pooled in a downstream processing (DSP) reactor. This reactor purifies the magnetic beads, removing residual contaminants such as enzymes, DNA strands, unused nucleotides, and buffer components. The result is a highly purified RNA drug substance, ready for final filtration.